What Was the Approximate Concentration of Solute in the Potato Tuber? How Do You Know
Potato Osmosis Lab
With schools around the earth shut downward, I know that y'all might exist scrambling for activities that can be done with students online. I have taken this classic biological science lab activity illustrating the principles of improvidence and osmosis and adjusted it as an online activeness. I did this lab many times with my 10th grade regular bio class at Kelly High School in Chicago, merely information technology can exist used successfully with kids ranging from center school to AP Bio. Students can read through the background here and make their own graphs, analyze these data, and depict conclusions.
I know you are working harder than ever before to teach your students despite the challenges you face. I promise you can utilize this activity or one of the others on this blog. Instructions are hither if you lot would like to create free access to DataClassroom for you lot students for 90-days. Stay well.
-Aaron Reedy

Groundwork
Molecules are constantly in motion as a issue of a prison cell'southward stored kinetic energy, which causes them to bump into each other and motility in random new directions. Diffusion is the motility of molecules from an area of where there are many (high concentration) to an area where there are fewer (low concentration). Osmosis is the diffusion of h2o through a semipermeable membrane. It is important to remember that a semipermeable membrane allows the solvent (usually h2o) to pass through, merely restricts the movement of a solute (a thing dissolved in the solvent). Water volition move across a semipermeable membrane from an area of lower solute concentration to an expanse of college solute concentration.
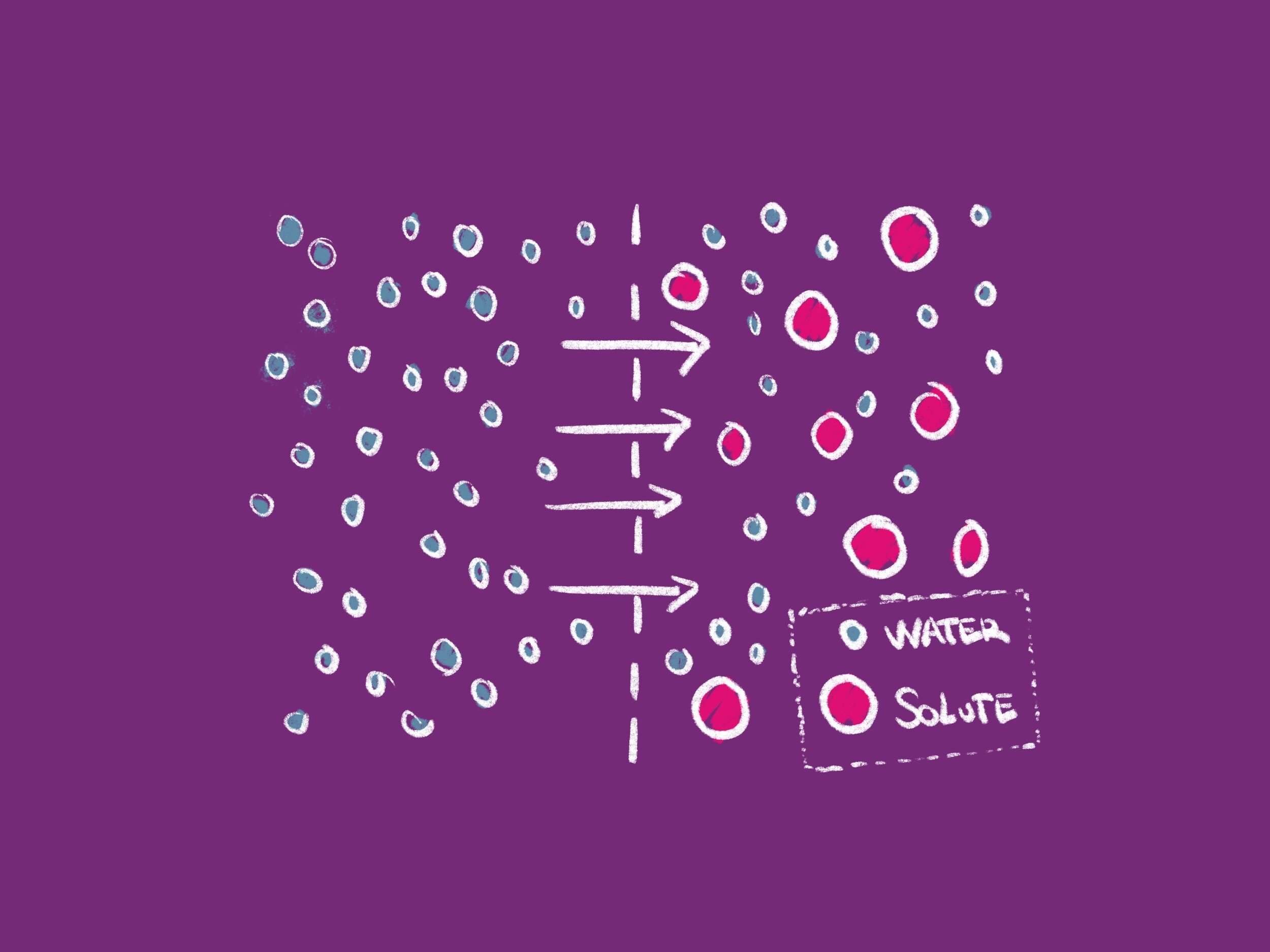
When each side of a membrane has equal solute concentration, the solution is said to be isotonic and h2o molecules will be as likely to motility in both directions across the membrane. In the case of a hypertonic solution, there is more solute exterior the cell than inside the cell. Hypertonic solutions cause water molecules to move out of the cell and into the region of higher solute concentration. Conversely, in hypotonic solutions at that place is a higher solute concentration inside the cell than outside, and water molecules move into the prison cell. Whenever possible, water will always move from an area of high water concentration/low solute concentration to an surface area of low water concentration/high solute concentration.
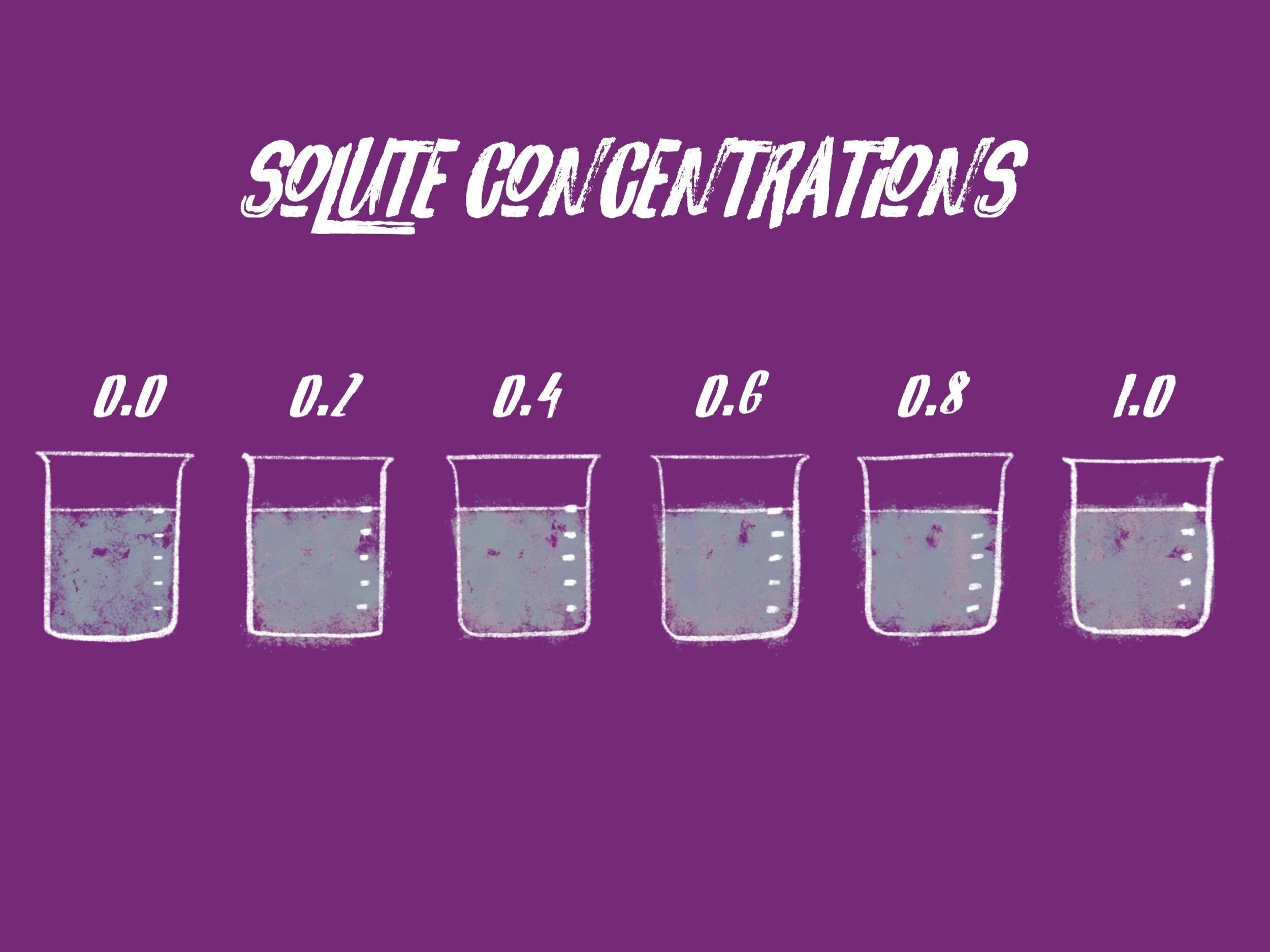
In this activeness, nosotros are going to explore osmosis by looking at a dataset produced with a archetype classroom experiment. The experiment uses pieces of potato that are placed in six dissimilar solutions of water each with a different solute concentration. The solute is sucrose and the concentrations are measured in units of molarity. The solutions range from no solute to a high concentration of solute and are 0.0 (distilled water), 0.2, 0.iv, 0.6, 0.8 and 1.0 molar sucrose.
Pieces of potato are cutting to similar sizes, weighed, and and then placed in ane of the six solutions overnight. The next day, the potato pieces are removed from the solutions, blotted dry, and their terminal masses are recorded.
Each row in this tidy dataset contains an observation for a single potato slice. Each column in the dataset is a variable and the cells in that cavalcade are the values of that variable. The variables recorded for each potato piece are Lab Grouping Proper noun, Sucrose Concentration (Molarity), Initial Mass (thousand), Final Mass (g), and Mass Change (%).
To come across a video conspicuously illustrating and explaining the general process for this lab, spotter Paul Andersen'south Bozeman Science video walkthrough:
The action
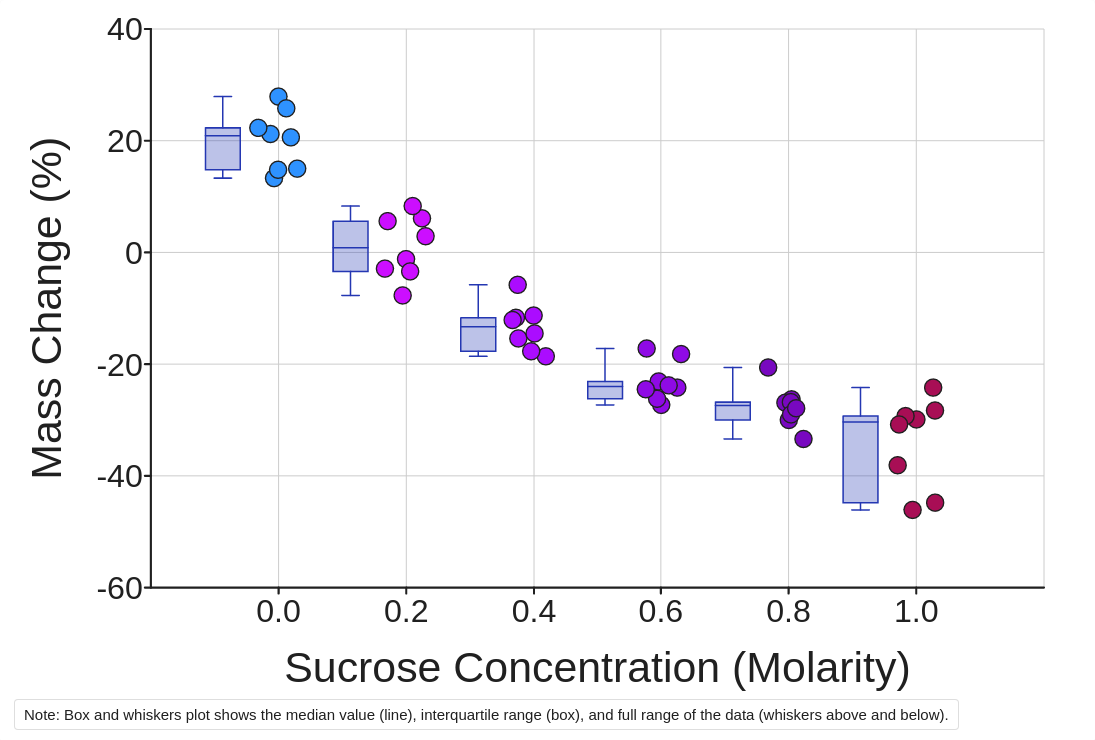
i. Click the xanthous Brand a graph button to visualize your information. Choose the scatter plot icon and Show Sucrose Concentration (Molar) on the 10-axis and Mass Change (%) on the Y-axis. You tin add descriptive statistics like means and medians by checking the box just to the right of the graph.
Find patterns in the data:
ii. What are the independent and dependent variables in this experiment?
three. How does Change in Mass (%) change with Sucrose Concentration (Molarity)?
4. Which substance moved across the cellular membrane in this action? What is the specific proper noun of the movement in terms of this substance?
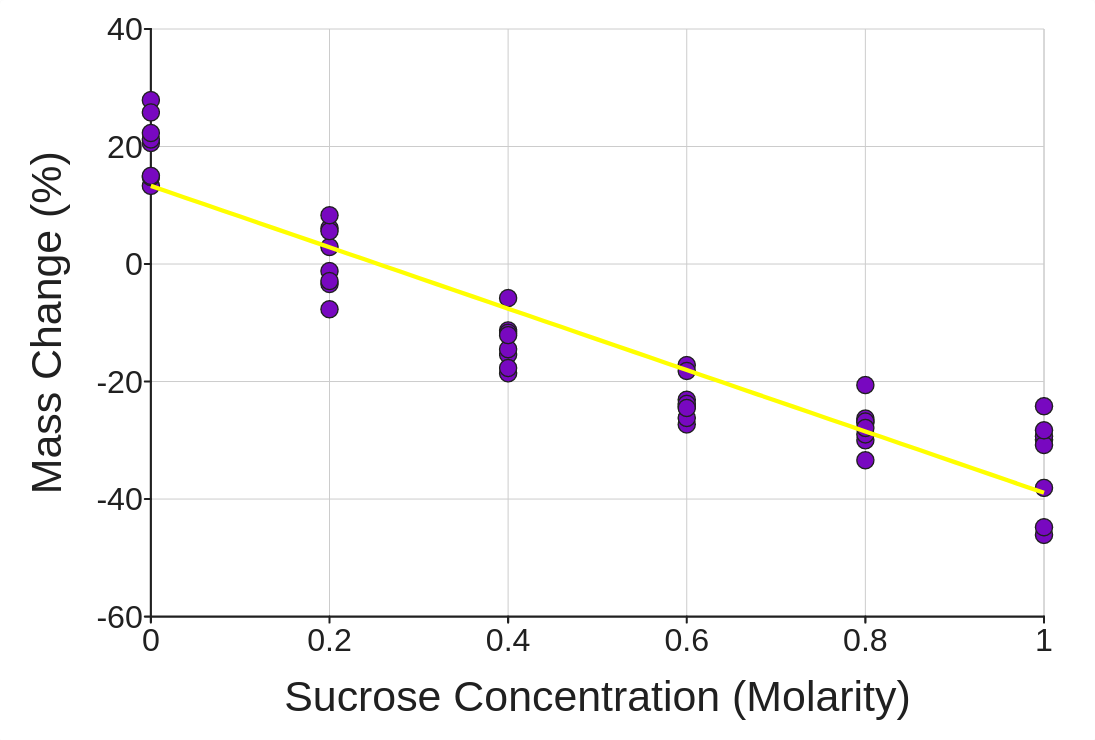
5. Now, change the variable called Sucrose Concentration (Molarity) to a Numeric variable with the dropdown menu right below the variable proper name near the top of the folio. Then add it dorsum to the graph once again by clicking the Show push button. Finally, add a regression line of best fit by checking the box just to the correct of the graph.
What is your all-time judge for the natural solute concentration inside a potato prison cell? Explicate how your information is bear witness for that judge.
six. Which solution is closest to being isotonic with respect to a potato cell? Which solutions were hypertonic/hypotonic? How do you know?
Challenge question:
vii. Using the principles illustrated with these data, explain why y'all tin can't drink seawater when lost at bounding main.
For a quick explanation of diffusion and osmosis, nosotros highly recommend Paul Andersen's AP Biological science Lab 1: Diffusion and Osmosis video. The explanation of the potato lab starts at 5:36.
Answer fundamental available to teachers upon request. Electronic mail info@dataclassroom.com
levinsonspeargons.blogspot.com
Source: https://about.dataclassroom.com/ready-to-teach/potato-osmosis-lab
0 Response to "What Was the Approximate Concentration of Solute in the Potato Tuber? How Do You Know"
Post a Comment